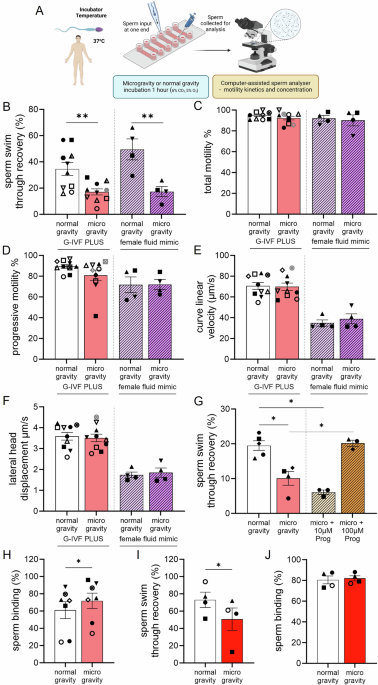
Study reports simulated microgravity impairs mammalian reproduction: human and mouse sperm show significantly reduced channel navigation (human P=0.002 and P=0.007 for media types), mouse fertilization rates fell ~30% after 4 h microgravity exposure (P=0.03), and pig fertilization and blastocyst formation were reduced (fertilization P=0.01; blastocyst formation P=0.005). Prolonged 24 h microgravity exposure delayed early cleavage and reduced blastocyst cell numbers in mice (epiblast cells reduced P=0.04; trends for lower total and ICM cells P≈0.05–0.06), while 100 µM progesterone restored human sperm navigation under microgravity and embryos that formed often had higher ICM/epiblast allocation. Relevance is high for space life‑support and reproductive biotech R&D but presents minimal immediate market impact.
This line of research creates a durable, non-consumer demand vector for clinical-grade culture systems and qualified consumables — think recurring revenue rather than one-off instrument sales. Procurement cycles for space agencies and defense contractors are long but predictable: expect initial qualification contracts and standard-setting pilots within 6–18 months, with meaningful purchasing (and follow-on clinical translations) in a 12–36 month window. Vendor concentration is the main structural risk: if a single proprietary clinostat or assay becomes the de facto standard, gatekeeping can capture margin and slow broader adoption unless competitors certify against that standard quickly. The highest-probability commercial outcome is modest re-specification of existing IVF and lab workflows (higher QA, traceable media, validated time-lapse systems) rather than a radical technology shift — that favors large diversified lab-capex and consumable vendors over one-product microgravity specialists. Second-order beneficiaries include makers of microfluidic slides, qualified gases/incubators, and regulated single-use disposables that can attach to long-term service contracts. Conversely, pure-play small caps that bet on one proprietary clinostat or a single-spaceflight validation without a diversified revenue base are exposed to binary downside if agencies or clinics standardize elsewhere. Catalysts to watch: publication of follow-up spaceflight validation (real microgravity) and procurement awards by agencies (6–24 months), and regulatory guidance on “space-grade” clinical protocols (12–36 months). Reversal risks are clear — a failed spaceflight validation, major reproducibility issues, or an ethical/regulatory clampdown could collapse the nascent demand curve and relegate this back to niche academic research.
AI-powered research, real-time alerts, and portfolio analytics for institutional investors.
Request a DemoOverall Sentiment
neutral
Sentiment Score
0.00
Ticker Sentiment