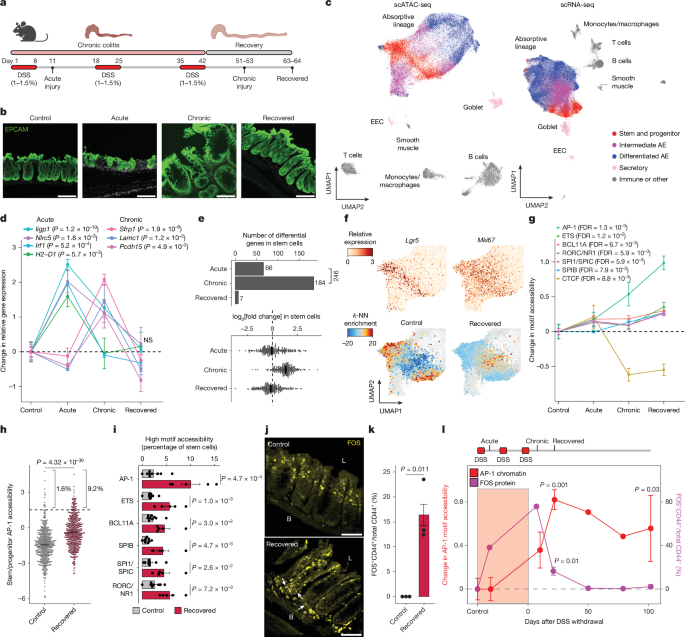
Epigenetic memory of colitis in mouse colonic stem cells persists for >100 days and is marked by cumulative AP-1 transcription factor accessibility, with ~9–12% of stem cell clones showing exceptionally high AP-1 accessibility. This clonal, heritable epigenetic state primes cells for accelerated early tumour outgrowth after APC loss (higher fraction of oversized microscopic lesions: 8.7% vs 2.5%) and AP-1 inhibition during tumor initiation reduced median tumour size by ~40%. The study introduces SHARE-TRACE (simultaneous single-cell gene expression, chromatin accessibility and clonal tracing) and suggests diagnostic and therapeutic opportunities to track or disrupt inflammation-driven epigenetic priming in chronic inflammatory disease.
This paper creates a clear demand vector for the upstream life‑science stack: specialized long oligos (nuclear retention motifs, split‑pool barcoding), robust methylation and low‑input library kits, and extracellular matrix/consumables for sustained organoid expansion. Expect a multi‑year step‑change in per‑sample reagent intensity versus bulk sequencing workflows — single‑cell + lineage + spatial assays consume 2–5× the reagent/consumable budget of a standard WGS/RNA run and drive recurring revenue. A second‑order commercial effect: translational programs will shift from mutation‑centric surveillance to epigenetic field mapping, creating recurring demand from surveillance/regulatory pathology labs if payors accept predictive epigenetic risk markers. That makes companies with turnkey methylation/extraction workflows and clinical‑grade sample prep (not just research tools) strategic beneficiaries. Risks and timing: clinical qualification, reimbursement and prospective validation are the gating factors — realistic commercialization is 12–36 months for research‑use expansion and 3–6 years to meaningful clinical revenue. Reversal catalysts include negative human validation, cheaper bulk proxies (improved cfDNA epigenetics) or regulatory setbacks for methylation‑based risk tests. On the therapeutic side, AP‑1 as a target looks druggable but will face the usual oncology safety and target‑engagement hurdles; expect small molecule or biologic efforts to take multiple years to read out in the clinic.
AI-powered research, real-time alerts, and portfolio analytics for institutional investors.
Request a DemoOverall Sentiment
neutral
Sentiment Score
0.00
Ticker Sentiment