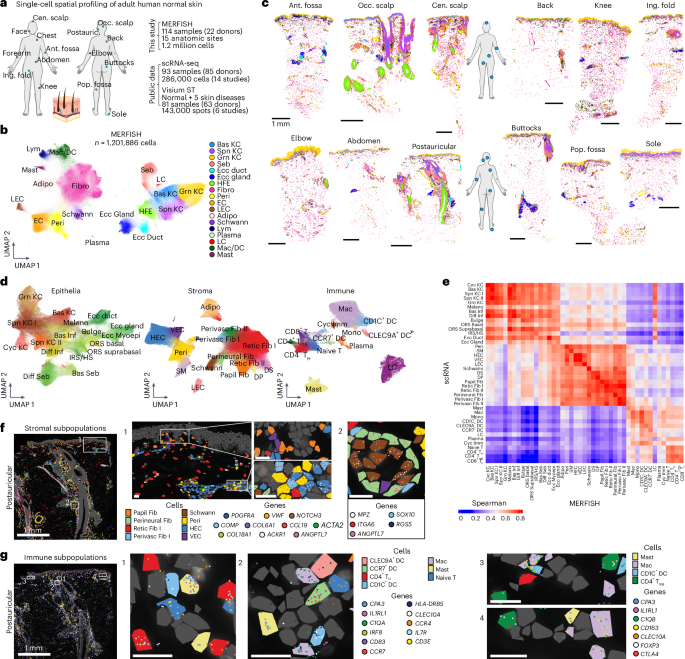
The authors built a 1.2 million-cell single-cell spatial MERFISH atlas from 114 samples across 15 anatomic sites and 22 donors, resolving 45 cell populations and 10 recurrent multicellular neighborhoods. They identify an immune-enriched perivascular neighborhood (PERIVASC I) where TNF sustains CCL19+ perivascular fibroblasts and show PERIVASC I expansion and transcriptional remodeling across multiple skin diseases (AD, psoriasis, HS, BCC, SCC). Processed data and an interactive webtool are provided for follow-up; findings are biologically and translationally relevant but are unlikely to move markets.
This dataset materially de-risks a class of biological readouts that pharma historically treated as low-resolution (anatomic site and compartment-level measures). That creates a two‑tier downstream market: (1) durable demand for high-throughput consumables and instrumentation from large life‑science suppliers as spatial profiling moves from pilot to routine; and (2) a new cohort of translational endpoints that could shorten early proof‑of‑concept studies for dermatology immunomodulators by converting spatial architecture changes into quantifiable biomarkers within 6–24 months. Expect procurement patterns to shift from one‑off academic buys to centralized, recurring service contracts at large CROs and hospital systems, amplifying volumes for incumbents that can integrate imaging, wet‑lab, and informatics stacks end‑to‑end. The strongest second‑order commercial pressure is on molecular diagnostics and CDx strategy: drug developers will seek spatial assays to stratify patients and show mechanism‑of‑action, pressuring diagnostic vendors to supply CLIA‑compatible, low‑variance panels and pathologist workflows. That favors vertically integrated platform vendors that already sell reagents, instruments and enterprise software, and creates an opening for partnerships between instrument OEMs and mid‑cap molecular diagnostics firms to co‑develop dermatology CLIA assays within 12–36 months. Regulatory and reimbursement timelines are the main gating factors — if payers demand hard clinical benefit rather than tissue‑architecture surrogates, adoption will stall; conversely, early adoption by high‑value specialty trials (dermatology, oncology) could fast‑track clinical validation. Short‑term risks (0–12 months) include vendor competition over proprietary chemistries and a fragmented standards environment that slows enterprise sales; medium term (12–36 months), the key reversal risk is failure to correlate spatial signatures with durable clinical endpoints across multiple patient cohorts. Monitor three binary catalysts: (A) major pharma announces spatial assay as primary or stratification biomarker in a Phase II program; (B) a large CRO or reference lab signs a multiyear supply/service agreement with a platform vendor; (C) payer guidance publishes on use of spatial biomarkers. Each would likely re‑rate tool vendors and specialist diagnostics providers by 10–30% upon public disclosure.
AI-powered research, real-time alerts, and portfolio analytics for institutional investors.
Request a DemoOverall Sentiment
neutral
Sentiment Score
0.00
Ticker Sentiment