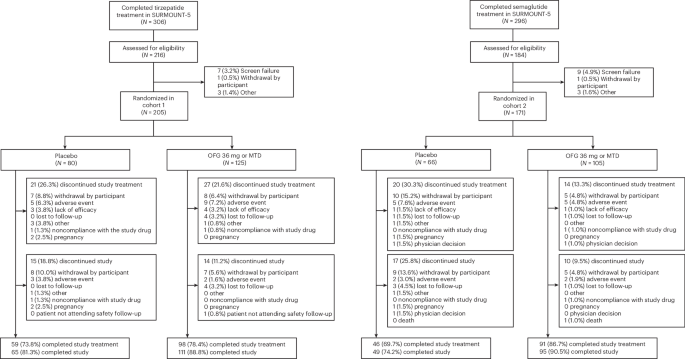
Eli Lilly’s phase 3b ATTAIN-MAINTAIN trial showed once-daily oral orforglipron preserved 74.7% of prior weight loss after tirzepatide and 79.3% after semaglutide at week 52, versus 49.2% and 37.6% with placebo, both with P < 0.001. The drug also maintained cardiometabolic improvements, with mostly mild-to-moderate gastrointestinal adverse events and no new liver safety signal. The results support orforglipron as a scalable oral option for long-term obesity treatment and could strengthen the drug’s commercial profile.
This is less about incremental efficacy and more about preserving the economics of the obesity franchise as the market shifts from initiation to chronic maintenance. The key second-order readthrough for LLY is that an oral GLP-1 with acceptable transition tolerability lowers the friction of persistence, which should expand the addressable pool beyond injection-tolerant patients and reduce the share of demand lost to discontinuation after the first year. That matters because maintenance is where lifetime value compounds; if the oral option becomes the default “step-down” or “convenience” product, it can keep patients inside Lilly’s ecosystem even if they later de-escalate from injectable therapy. The competitive implication is asymmetrical versus Novo: this data reduces the moat of injectables as the only credible long-duration obesity solution and makes formulation/route-of-administration a more important battleground than pure efficacy deltas. A successful oral maintenance strategy also improves payer optics because it gives plans a cheaper, more scalable continuation pathway after initial weight loss, potentially supporting broader coverage rather than tighter prior authorization. That could pressure emerging oral competitors that lack either the same brand credibility or a clean transition story from existing GLP-1 users. The main risk is that the market may over-rotate to the “oral is enough” narrative before we see head-to-head durability versus continued injectables and before real-world adherence data validate the trial’s transition assumptions. The biggest reversal catalyst is evidence that oral persistence in practice is poor once GI burden, daily dosing, and payer switching friction hit real patients over 6-12 months. In the near term, the article is bullish for LLY sentiment; over 12-24 months the real variable is whether this becomes a maintenance bridge product or simply an underwritten convenience layer that cannibalizes higher-margin injectable growth.
AI-powered research, real-time alerts, and portfolio analytics for institutional investors.
Request a DemoOverall Sentiment
moderately positive
Sentiment Score
0.62
Ticker Sentiment