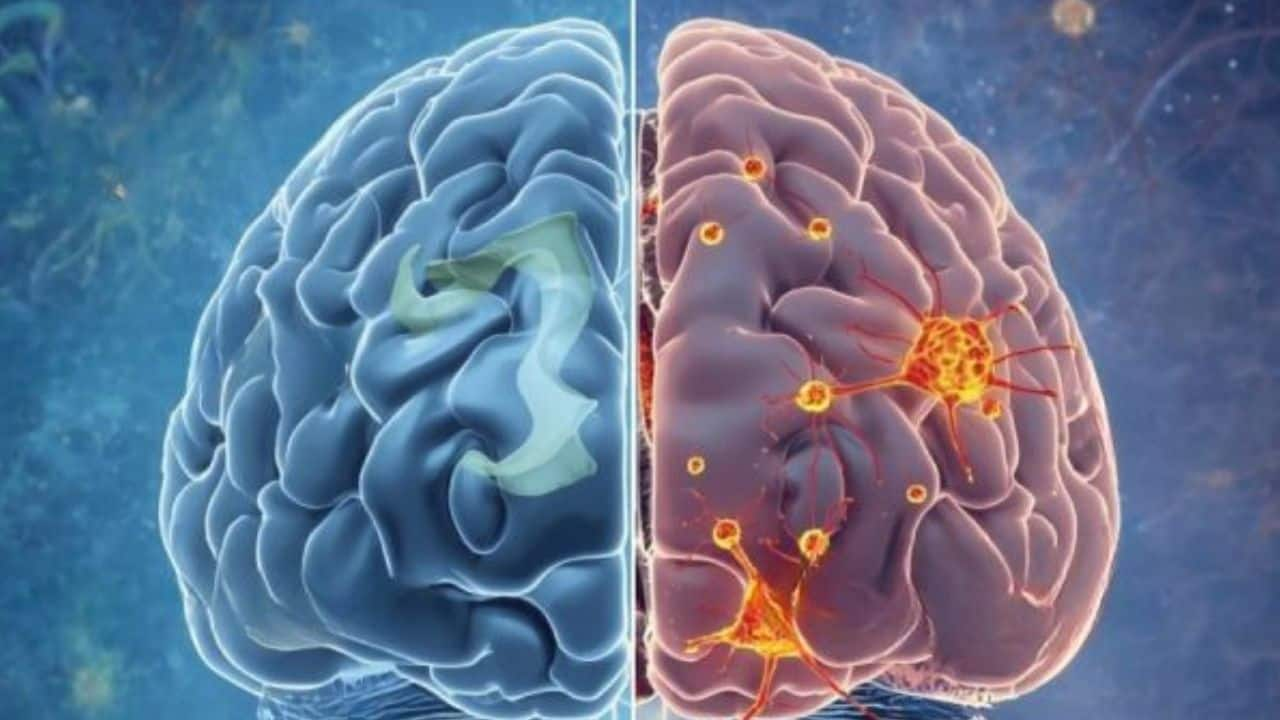
Heidelberg University researchers identified a TRPM4–NMDA 'death complex' that drives nerve‑cell loss in Alzheimer’s-model mice and showed the experimental compound FP802 disrupts this complex, reducing synaptic loss, mitochondrial damage, cognitive deficits and amyloid‑β levels. Results are preclinical (mouse models); significant safety studies and human trials are required, so near‑term market impact is limited but the finding establishes a new therapeutic target that could shape Alzheimer’s drug development.
The key market implication is that a credible, druggable downstream node in neurodegeneration shifts value away from single-modality amyloid-clearing plays toward platform owners and service providers who can accelerate IND-enabling programs. If the signal replicates in human tissue and shows a measurable CSF or PET biomarker response, expect a concentrated 18–36 month window where small academic spinouts become buyout targets; acquirers will pay a premium for assets that materially reduce time-to-proof-of-concept in symptomatic patients. Translational risk is the dominant macro hedging parameter: CNS programs historically suffer >70% attrition between preclinical efficacy and Phase 2. Two specific failure modes matter more here — target engagement not measurable in humans, and off-target safety (cardiac/neuromuscular) driven by broadly expressed ion channels — both can invert expected value within 6–18 months after first-in-human dosing. Conversely, a clean Phase 1 biomarker update showing target engagement plus a downstream mitochondrial/neuronal-synapse signal would re-rate the space quickly. Second-order beneficiaries are non-obvious: specialized CNS CROs/CDMOs and PET tracer developers (biomarker infra) that can turn a mechanistic readout into a regulatory path to Phase 2. Big pharmas with late-stage AD franchises or M&A firepower will behave as strategic buyers rather than bidders for platform equity, compressing time-to-liquidity but widening the valuation gap between platform owners and single-asset speculative biotechs. Monitor IND filings, human tissue replication, and first human biomarker readouts as primary catalysts over the next 12–36 months.
AI-powered research, real-time alerts, and portfolio analytics for institutional investors.
Request a DemoOverall Sentiment
mildly positive
Sentiment Score
0.25