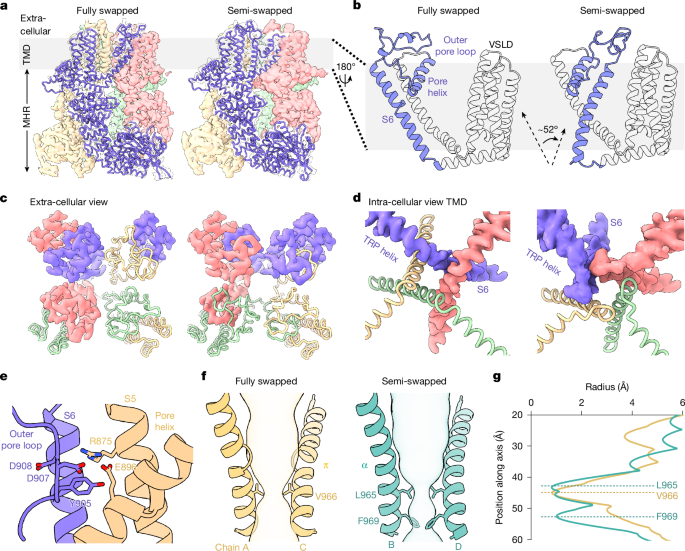
Study defines structural and energetic mechanism for TRPM8 cold activation: TRPM8 is activated below ~26 °C and menthol binding stabilizes the TRP helix by ~1.4 kcal·mol−1. HDX–MS shows bimodal populations in pore and TRP helices with >3 kcal·mol−1 folding-energy differences and a ~52° bending/reorientation of S6 in a newly identified ‘semi-swapped’ architecture. Findings explain species differences (avian versus human) and implicate outer-pore/PIP2 interactions as druggable regulatory nodes, relevant for therapeutic targeting of thermosensory channels.
Recent convergence of membrane-native structural capture with residue-level thermodynamic readouts raises the bar for ion-channel target validation and should materially shorten early R&D cycles for programs that previously required iterative mutagenesis and low-throughput functional mapping. For instrument and consumables vendors this is a demand-shift from one-off hardware sales to recurring high-margin reagents, bespoke lipids and bespoke sample-prep workflows — an install can produce steady consumable pull-through for years rather than a single capital sale. Competitive dynamics favor vendors with end-to-end capabilities (cryo front-end, sample prep, mass spec analytics and software) because pharmaceutical groups will prefer single-supplier validated workflows to minimize reproducibility risk; mid-sized niche players that supply standardized native-lipid libraries or HDX automation could become attractive acquisition targets. Expect pharma/biotech to accelerate collaborations and licensing deals around validated channel-targets once tertiary/energetic mechanisms are demonstrated; these deals are high-impact binary catalysts (partner announcement → program advancement) on a 6–24 month cadence. Principal tail risks: slower conversion from structural insight to clinic than investors expect (2–6+ years typical for ion-channel therapeutics), and capex sensitivity — instrument order cycles compress sharply in macro slowdowns and can wipe near-term revenue. Watch three short-term readouts as catalysts: large pharma validation partnerships, recurring consumable bookings in quarterly instrument reports, and published replication/standardization studies; any negative surprise on these can reverse momentum quickly. Contrarian take: the market likely underestimates the value of recurring consumables and validated workflows; upside is concentrated in firms that can bundle hardware, software and consumables into auditable workflows. Conversely, the boost to basic discovery does not guarantee near-term drug royalties, so stock responses will be front-loaded to tool vendors rather than biopharma.
AI-powered research, real-time alerts, and portfolio analytics for institutional investors.
Request a DemoOverall Sentiment
neutral
Sentiment Score
0.00
Ticker Sentiment